- Home
- Services
- About
- News
- Contact
- Mdaemon bittitan
- Top illusion games
- Indian passport tatkal
- Butterfly escape game download for pc
- How to watch jio cinema without jio number
- Beamng drive xbox one
- Ayyappan tamil archanai
- How to calculate ppm serial dilution
- Download dell sonicwall netextender
- Chrysler drb iii battery
How to calculate ppm serial dilution how to#
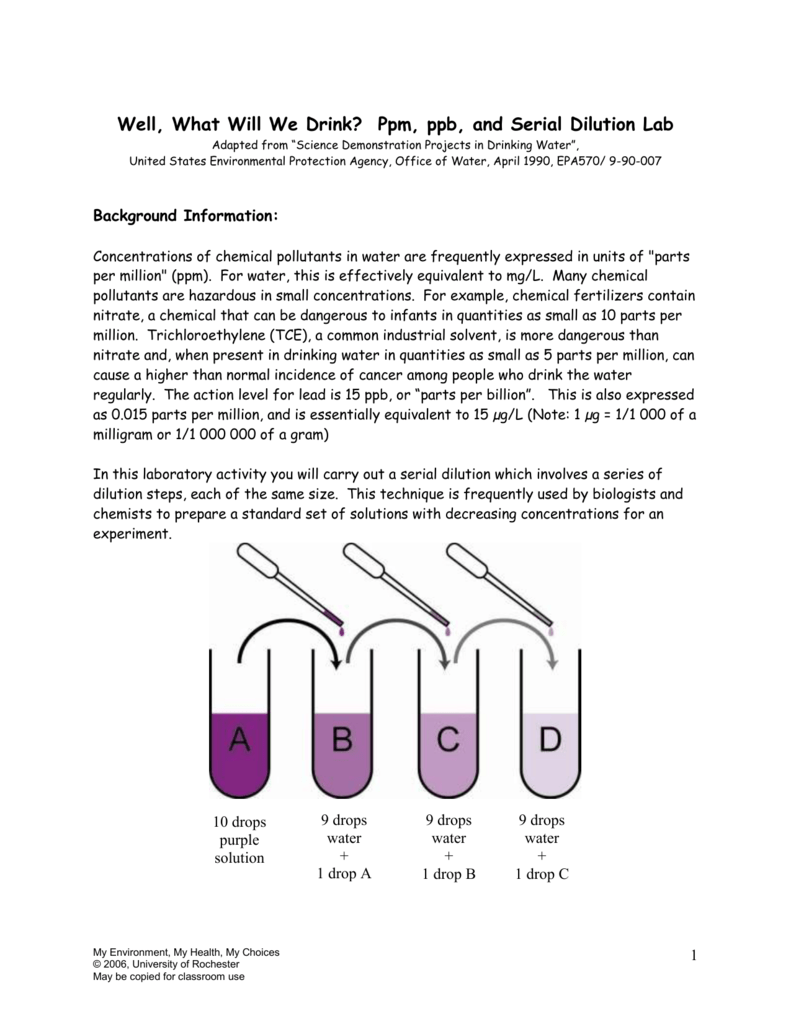
You will learn how to change % solution concentrations into ppm. To understand a little about really dilute solutions we offer the following liquid dilution investigation. Liquid solutions can also be described in ppm. After constructing the model and proving to yourself that is really does show 1 ppm you will be able to solve other concentration problems, both mathematically and conceptually. To accomplish this, you will create a life-sized cubic model to feel, touch, probe and investigate.

For a 96-well microplate format, this number is typically 7 (if using a single column) or 11 if using a single row. Enter the number of times the serial dilution needs to be performed. This investigation will help you develop a conceptual understanding of the term, concentration. Enter the dilution factor for the serial dilution. Scientists use the term ’ parts per million’ because the concentration of CO2 is so very small.
In other words, for every 1 million molecules of gas molecules you could find exactly 360 CO2 molecules. 360 ppm), identifies the number of CO2 molecules relative to exactly 1 million molecules of gas mixture. This amount of CO2, reported as 360 parts per million (abbr.

In 2006 a typical sample of air contained about 360 molecules of CO2 for every one million total molecules of the air mixture (1). The term ’ concentration’ tells us this information. Since scientists want to know how CO2 (or any other greenhouse gas) affects climate they need to describe accurately with numbers how much of this gas is floating around in our atmosphere. How can such a small amount of CO2 cause such an effect ? In this article critical concepts on dilutions are clarified with illustrative calculations, along with the explanation of how to prepare 100 ppm solution. Greenhouse gases in our atmosphere, like carbon dioxide (CO2), are causing Earth to heat up significantly yet, they occur in only miniscule amounts.
- Home
- Services
- About
- News
- Contact
- Mdaemon bittitan
- Top illusion games
- Indian passport tatkal
- Butterfly escape game download for pc
- How to watch jio cinema without jio number
- Beamng drive xbox one
- Ayyappan tamil archanai
- How to calculate ppm serial dilution
- Download dell sonicwall netextender
- Chrysler drb iii battery
